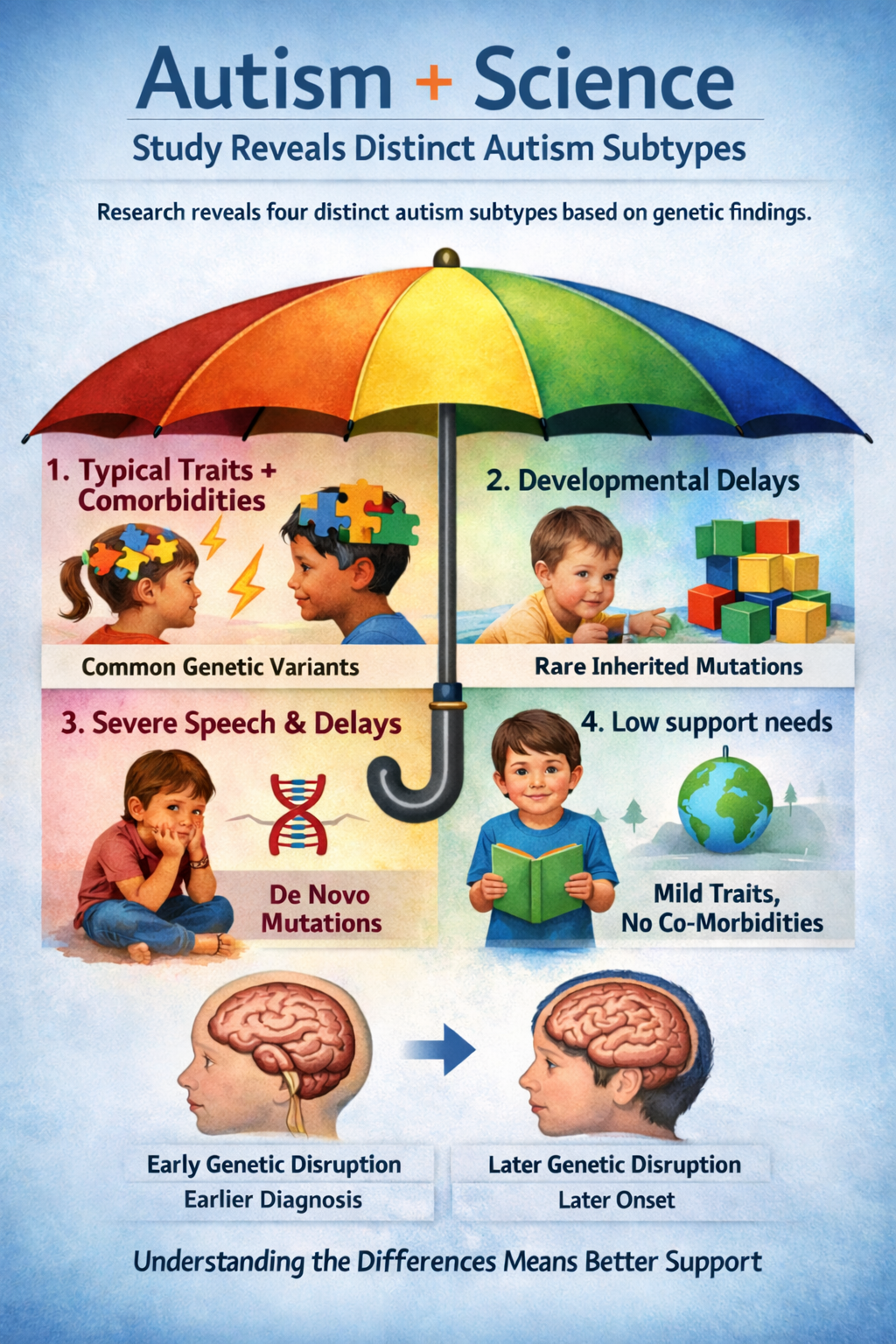
Progress is being made in understanding the part of the autism spectrum caused by genetic mutations. Researchers at Princeton University and the Simons Foundation analyzed data from over 5,000 autistic children and found four biologically distinct classes of autism, each with its own genetic architecture, co-occurring conditions, and developmental trajectory. Four different neurological realities. Four different sets of needs. One umbrella.
One subtype showed common autism traits such as repetitive behaviors and social challenges alongside conditions like ADHD and anxiety, driven largely by common genetic variants. Another was characterized by developmental delays tied to rare inherited mutations. A third displayed the greatest challenges with speech and more pronounced developmental delays, linked to high-impact spontaneous (de novo) mutations in highly constrained genes. The fourth group typically meet milestones at the same time as neurotypical peers, they do not usually have co-occurring psychiatric conditions, and they present more moderate core autism related behaviors.
The study also showed that the timing of when affected genes are expressed during brain development aligned with each class's clinical milestones. Children whose gene disruptions occurred in fetal stages showed earlier diagnoses and greater delays, while those with postnatal gene disruptions showed subtler, later-emerging profiles.
If the science recognizes that one subtype is driven by common genetic variants tied to ADHD and anxiety, while another carries rare inherited mutations linked to developmental delays, then support systems that treat these individuals identically aren't just inefficient, they're inequitable.
The science is becoming more precise. Now employers, policymakers, educators and healthcare providers need to do the same.